About author :
Kalpen N Patel*, Maulika S Patel
Shree Krishna Institute of Pharmacy, Shankhalpur, Becharaji, Mehsana, India
*e-mail :[email protected]
INTRODUCTION:
The aim of the present investigation is to formulation development and evaluation of pulsatile drug delivery system for Anti-Asthmatic drug of Montelukast sodium. Sodium CMC generally used for super disintegrator, it increased the lag time of pulsatile drug delivery system with decreasing the amount of quantity and increased the amount of quantity of Carbopol 971P was generally used for swelling agent, Eudragit RS PO is an enteric polymer and the pH of that 7.4. The formulations were evaluated for Drug content, in-vitro dissolution study, Disintegration time and lag time. The developed formulations of pulsatile drug delivery system which give the better lag time. From these formulation the optimize formulation F3 provide lag time of rupture of PRTs is 6:10 hr and release drug after rupture of outer coat. So we can conclude that the Formulation F3 give better lag time of rupture.
Keywords: Montelukast Sodium, Eudragit RS PO, Carbopol 971P, in-vitro dissolution study, lag time, drug content.
Description of Controlled Drug Delivery System:
Controlled drug delivery systems have acquired a centre stage in the area of pharmaceutical R & D sector. Such systems offer temporal &/or spatial control over the release of drug and grant a new lease of life to a drug molecule in terms of controlled drug delivery systems for obvious advantages of oral route of drug administration. However, there are certain conditions, which demand release of drug after a lag time. Such a release pattern is known as “Pulsatile release”. Due to advances in chronobiology, chronopharmacology and global market constraints, the traditional goal of pharmaceutics is becoming obsolete. The diseases currently targeted for chronopharmaceutical formulations are those for which there are enough scientific backgrounds to justify ChrDDS compared to the conventional drug administration approach. These include asthma, arthritis, duodenal ulcer, cancer,cardiovascular diseases, hypercholesterolemia, ulcer and neurological diseases. Chronopharmacotherapy for rheumatoid arthritis has been recommended to ensure that the highest blood levels of the drug coincide with peak pain and stiffness.
Description to Pulsatile Drug Delivery System :
A Pulsatile drug delivery system is characterized by a lag time that is an interval of no drug release followed by rapid drug release.
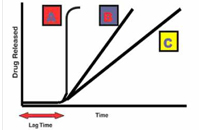
Figure: 1: Drug release profile of pulsatile drug delivery system
A: Ideal sigmoidal release, B & C: Delayed release after initial lag time
The first pulsed delivery formulation that released the active substance at a precisely defined time point was developed in the early 1990s. In this context, the aim of the research was to achieve a so-called sigmoidal release pattern (pattern A in Figure 1). The characteristic feature of the formulation was a defined lag time followed by a drug pulse with the enclosed active quantity being released at once. Thus, the major challenge in the development of pulsatile drug delivery system is to achieve a rapid drug release after the lag time. Often, the drug is released over an extended period of time (patterns B & C in Figure 1). This following reviews the various pulsatile drug delivery systems that are reported.
CONCLUSION:
The Aim of this study to developed time dependent pulsatile release tablets (PRTs) for treatment of asthma was satisfactory attempt using pH dependent polymer (drug coat S100) and evaluated in-vitro characterization studies. From FT-IR study and physical observation it can be concluded that there is no significant Drug-Excipients interaction. So we can conclude that drug and other excipients are compatible with each other. Appropriate factorial design can be successfully used in development of formulations based on drug coat S100 to achieve drug release in upper portion of large intestine. The lag time of PRTs increasing with increasing amount of carbopol 971P, levels of coating and decreased amount of Na.CMC. So lag time could be adjusted and modified by varying level of coating layer and amount of swelling materials. The optimize formulation F3 (Polymer ratio 3:1 and 10% drug coat S100 coating) provide lag time of rupture of PRTs is 6:10 and release drug after rupture of outer coat. So we can say that the Optimized Formulation F3 produce better lag time of rupture.
 Pharma Research Library | Pharma Info Index Research | Innovation | Opportunities |
Pharma Research Library | Pharma Info Index Research | Innovation | Opportunities |


